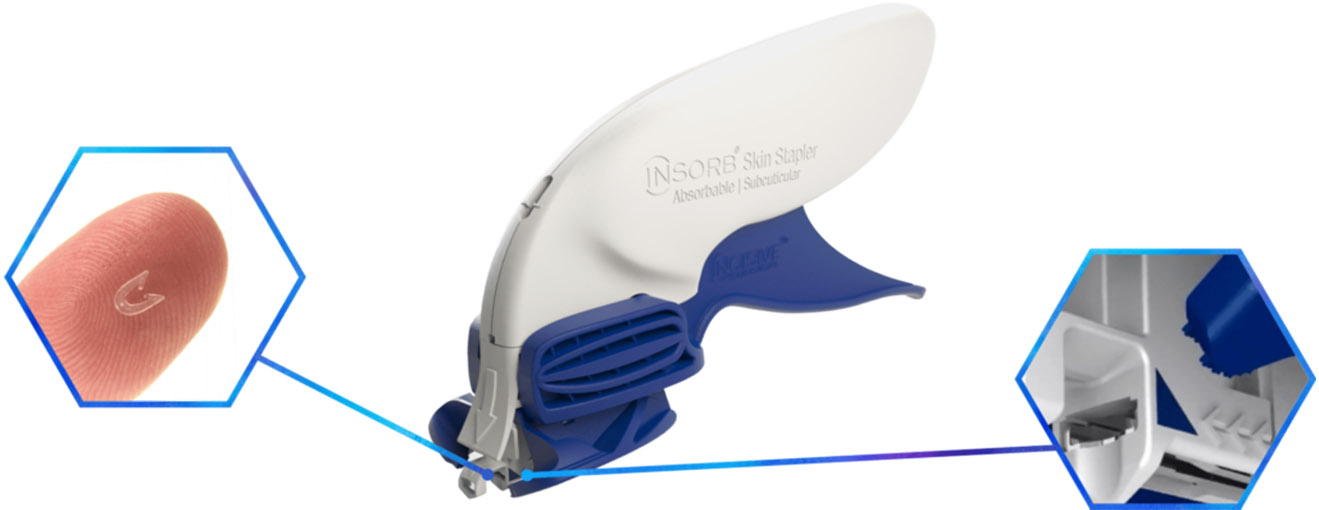
INSORB™ Absorbable
Skin Stapler
From INSORB® Absorbable Skin Stapler part of Women’s & Family Care

The Rapid, Cosmesis-Proven Skin Closure Solution1,2,3
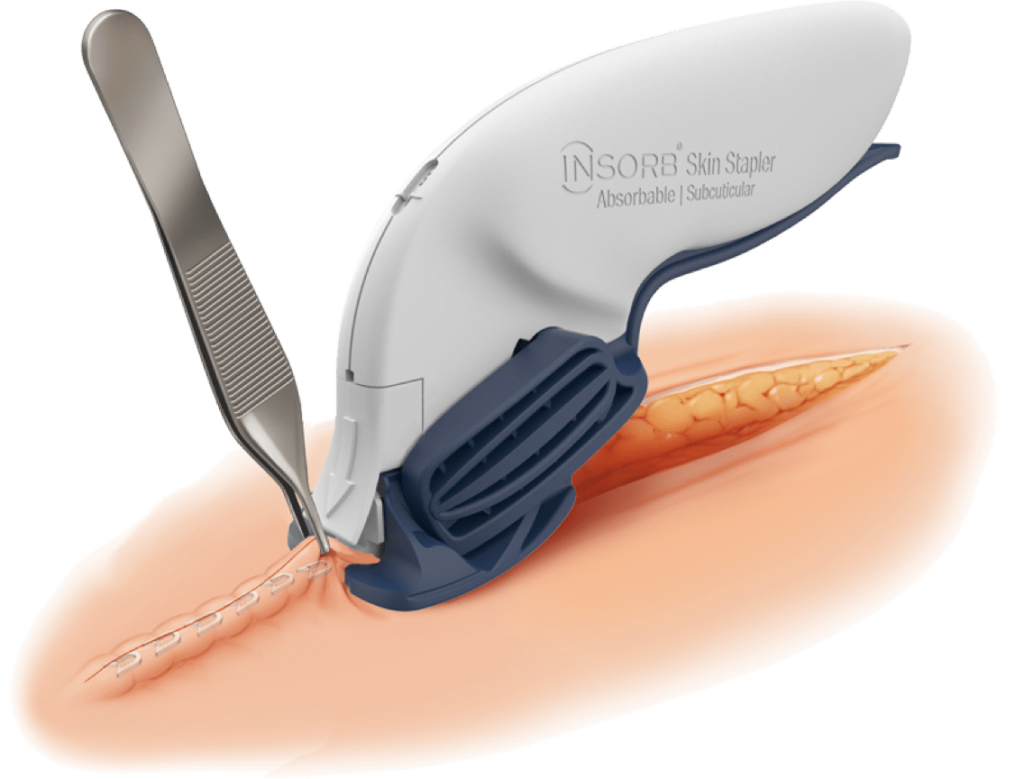
The INSORB™ Absorbable Skin Stapler is a subcuticular, interrupted closure modality designed to create uniform, symmetric dermis to-dermis closures.
- Increases skin eversion, which may improve healing and cosmesis2,4
- Reduce closure time compared to sutures8,1,*
- Eliminates the percutaneous insult created by metal skin staples5,6,†
The Staple
The INSORB Absorbable Skin Staple is composed of a co-polymer derived from polylactide-polyglycolide which has a well-established6 history in wound closure.
INSORB Stapler
The patented INSORB Skin Stapler is a sterile, single patient use device that deploys 30 absorbable staples, sufficient to close an incision up to 21 cm in length, when placed at 7 mm intervals.
Blue Triangles
Keep the blue triangles fully covered by the tissue. This will assist with proper alignment of the staples and minimize external placements.
Why INSORB?
Benefits of INSORB vs. Suture
The INSORB absorbable skin stapler is designed to create uniform, symmetric dermis-to-dermis closures.
- Increases skin eversion, which may improve healing and cosmesis2,4
- Reduce closure time compared to sutures8,1,*
- May result in reduced hand fatigue8
- Eliminates risk of needlestick injuries at the time of closure5,9
- Less hypertrophic scarring found in breast surgery10,‡
Benefits of INSORB vs. Metal Staples
The INSORB absorbable skin stapler eliminates the percutaneous insult created by metal skin staples.5,6,†
- Prevents the potential of “railroad track” scars5,6
- Evidence of reduced wound complications in c-sections11
- Spare patients inconvenience, anxiety, and discomfort associated with metal staple removal6,12
- Potentially less pain immediately post-op in C-Section13
Cosmetic Result of INSORBTM Absorbable Skin Staples
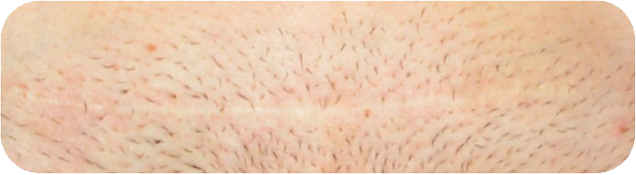
1 YEAR POST-OP
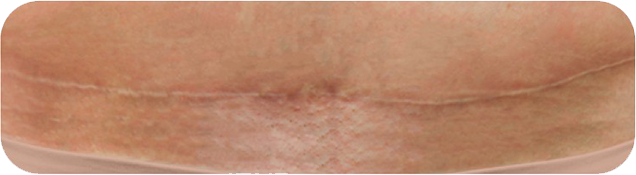
1 YEAR POST-OP
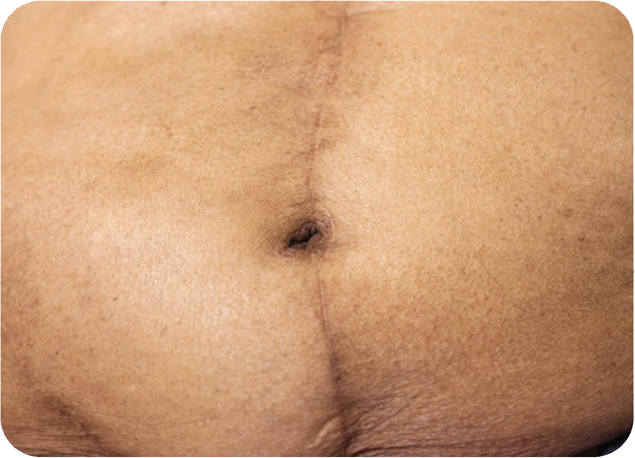
1 YEAR POST-OP
There are multiple factors that can affect patient results and recovery and these results may not be representative.

Learn more about the rapid, cosmesis-proven, skin closure solution1,2,3
INSORB Videos
INSORB Instructional Video
INSORB Images
Images of INSORB performance from multiple patients.
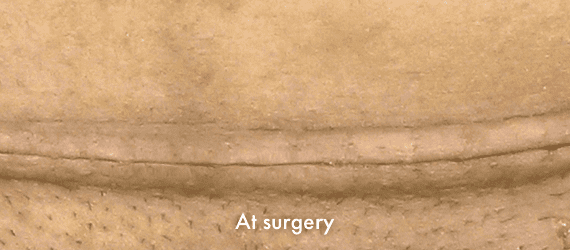
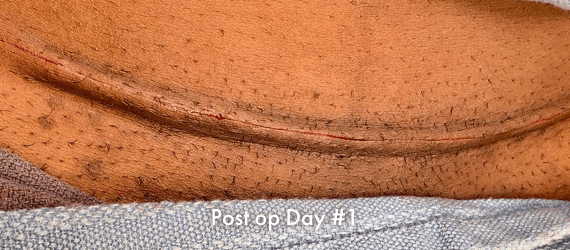
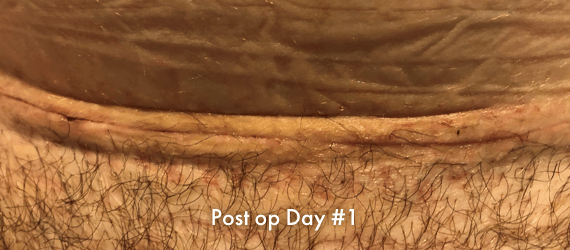
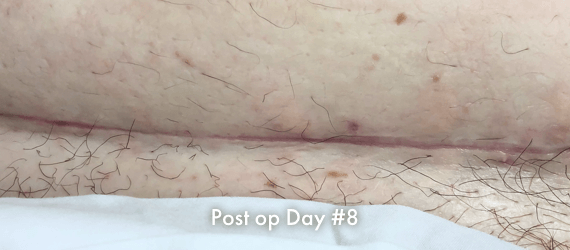
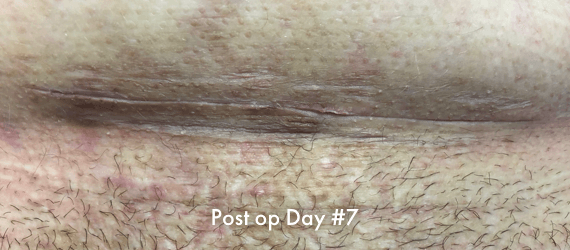
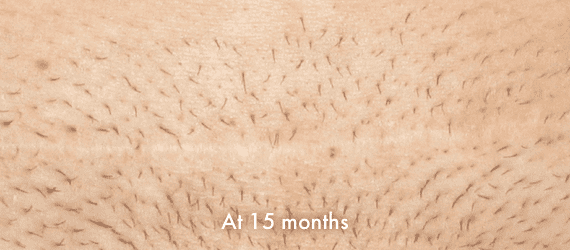
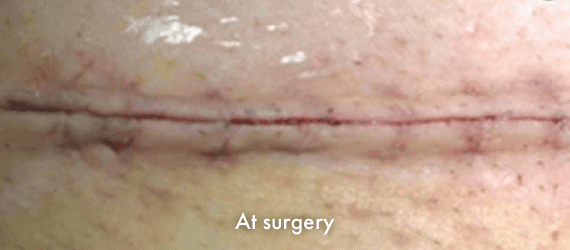

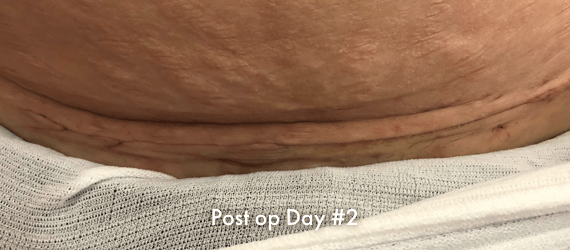

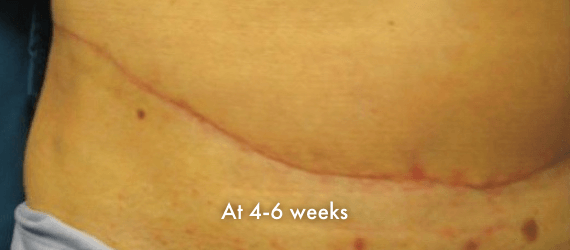
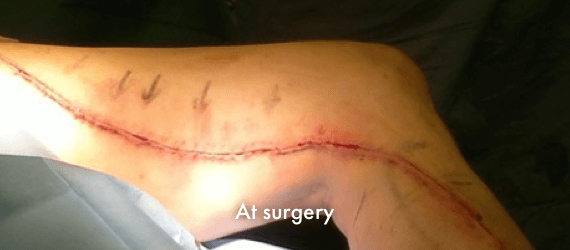
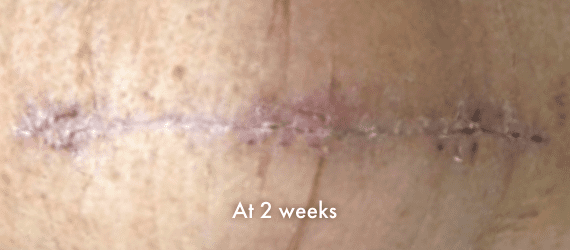
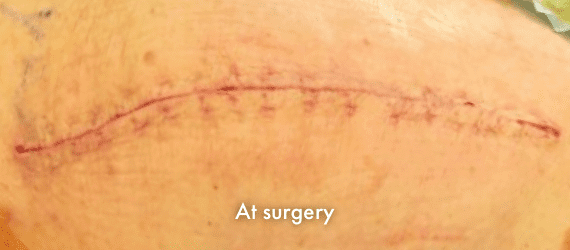
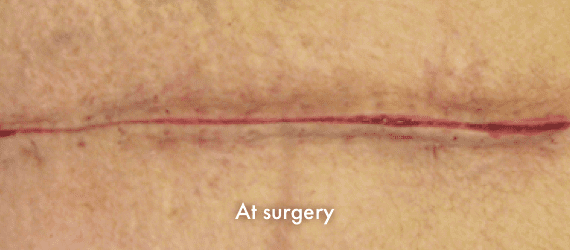


There are multiple factors that can affect patient results and recovery and these results may not be representative.
Documents
Product Brochure
Product Brochure
Product Catalogs
Product Catalogs
Product Codes
| Product | Order Code |
|---|---|
| INSORB Absorbable Skin Stapler – Box of 6 | 2030 |
| Double Adson Forceps | 9100 |
Footnotes
* Demonstrated in Abdominoplasty, Mammoplasty, and C-Section
† For proper technique, Insorb IFU
‡ Compared to suture 8.3% with INSORB vs. 20.7% with standard sutures in breast surgery
References
1. Madsen, Annetta Madeline, et al. Absorbable Subcuticular Staples Compared with Suture for Cesarean Closure. Obstet Gynecol. 2015; vol. 125
2. Cross, K. J., Teo, E. H., Wong, S. L., Lambe, J. S., Rohde, C. H., Grant, R. T., & Ascherman, J. A. (2009). The absorbable dermal staple device: a faster, more cost-effective method for incisional closure. Plastic and Reconstructive Surgery, 124(1), 156–162.
3. Patel, V., Green, J. L., Christopher, A. N., Morris, M. P., Weiss, E. S., Broach, R. B., & Butler, P. D. (2021). Use of Absorbable Dermal Stapler in Reduction Mammoplasty: Assessing Technical, Quality-of-Life, and Aesthetics Outcomes. Plastic and Reconstructive Surgery. Global Open, 9(8), e3784–e3784.
4. Kalaskar B.Tech PhD, Dr Deepak K., Butler MD FRCSI FRCS FRCS (Plast) Professor of Plastic and Reconstructive Surgery UCL, Professor Peter E., Ghali, Shadi: Plastic and Reconstructive Surgery. London, UK. 2016
5. INSORB Surgical Stapler [Instructions For Use]. Trumbull, CT. CooperSurgical, Inc.; 2019
6. Dresner, H. S., & Hilger, P. A. (2009). Comparison of incision closures with subcuticular and percutaneous staples. Archives of Facial Plastic Surgery, 11(5), 320–326.
7. Gunatillake P.A. and A. R. (2003). Biodegradable Synthetic Polymer for Tissue Engineering. European Cells and Materials, CSIRO Molecular Science. Volume 5.
8. Bron, T., & Zakine, G. (2016). Placement of absorbable dermal staples in mammaplasty and abdominoplasty: A 12-month prospective study of 60 patients. Aesthetic Surgery Journal, 36(4), 459–468.
9. The International Healthcare Worker Safety Center (University of Virginia Health System)
10. Caye, N. (2011). The benefit of the INSORB® subcuticular skin stapler in plastic surgery. Preliminary 2-year study in breast and abdominal surgery. Journal of Wound Technology, 14, 30-32.
11. Schrufer-Poland, T. L., Ruiz, M. P., Kassar, S., Tomassian, C., Algren, S. D., & Yeast, J. D. (2016). Incidence of wound complications in cesarean deliveries following closure with absorbable subcuticular staples versus conventional skin closure techniques. European Journal of Obstetrics & Gynecology and Reproductive Biology, 206, 53–56
12. Feese, C. A., Johnson, S., Jones, E., & Lambers, D. S. (2013). A randomized trial comparing metallic and absorbable staples for closure of a Pfannenstiel incision for cesarean delivery. American journal of obstetrics and gynecology, 209(6), 556.e1–556.e5565.
13. Nitsche, J., Howell, C., & Howell, T. (2012). Skin closure with subcuticular absorbable staples after cesarean section is associated with decreased analgesic use. Archives of gynecology and obstetrics, 285(4), 979–983. https://doi.org/10.1007/s00404-011-2121-5