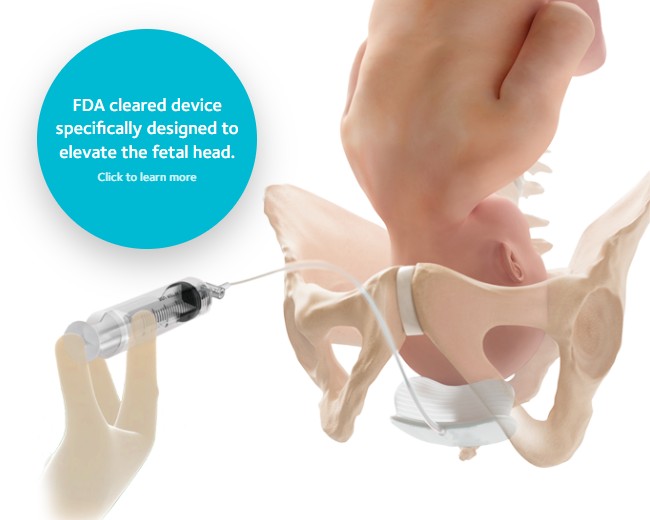
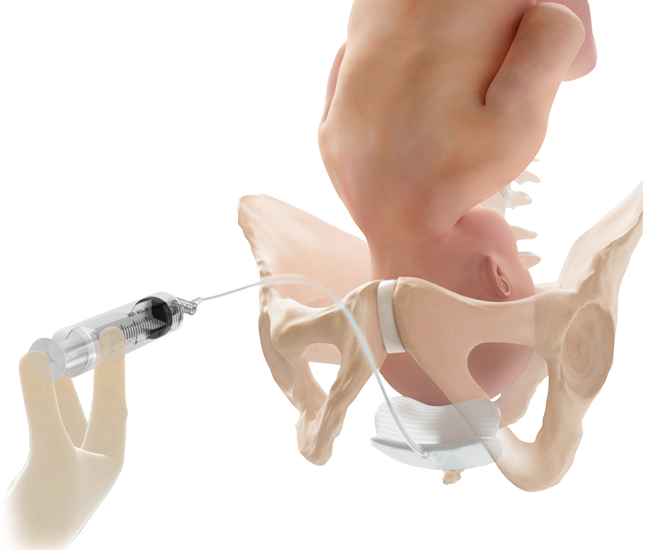
Fetal Pillow™ Cephalic
Elevation Device
Fetal Pillow is intended to elevate the fetal head and facilitate delivery of the fetus in women requiring a Cesarean section at full dilation or those requiring a Cesarean section after a failed instrumental vaginal delivery.1
Fetal Pillow is indicated for use in gestational age ≥37 weeks.
How Fetal Pillow Works1
The device follows a simple, standardized process designed to integrate into existing labor and delivery workflows.
Note: Before inserting, inflate the device with 60 cc saline prior to use to check the integrity of the device. Empty the device using the syringe provided before insertion.
1. Prepare & Insert
Fold the device and insert vaginally using sterile lubrication. Ensure balloon is in contact with the fetal head and base plate is in contact with the pelvic floor.
2. Correct Positioning
Once inserted, push the device posteriorly until it is touching the coccyx.
3. Patient Positioning
Patient’s legs are placed flat before inflation.
4. Inflation
A total of 180 cc of saline is required to produce the desired elevation.
5. Perform Cesarean Section
Proceed using standard surgical technique.
6. Deflation & Removal
Deflate after delivery and remove device vaginally.
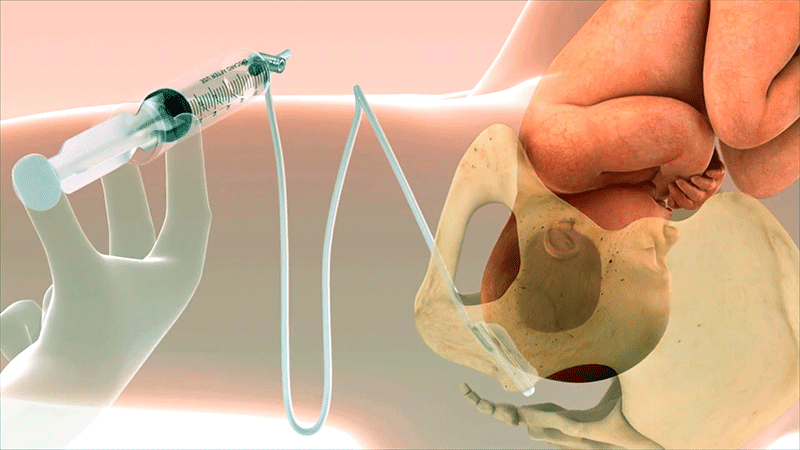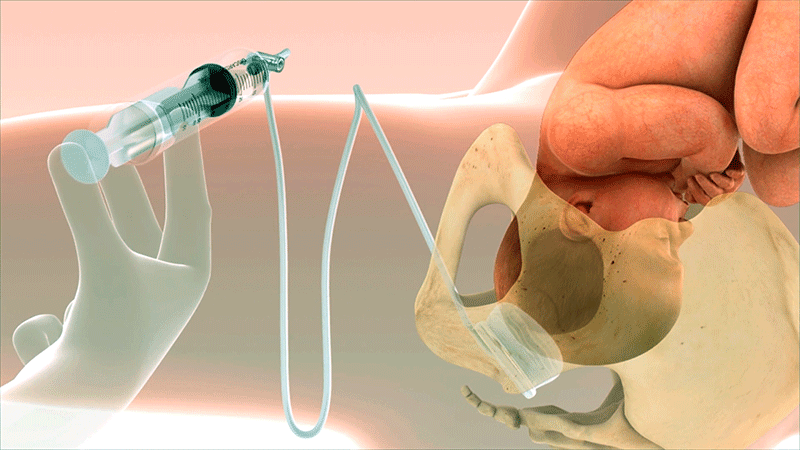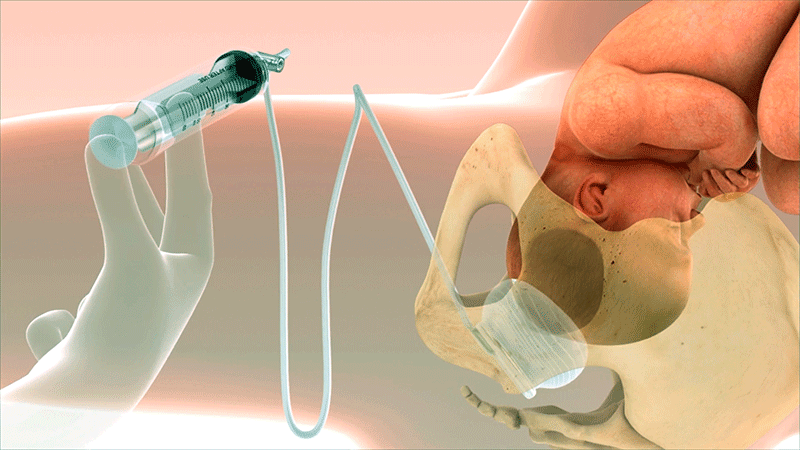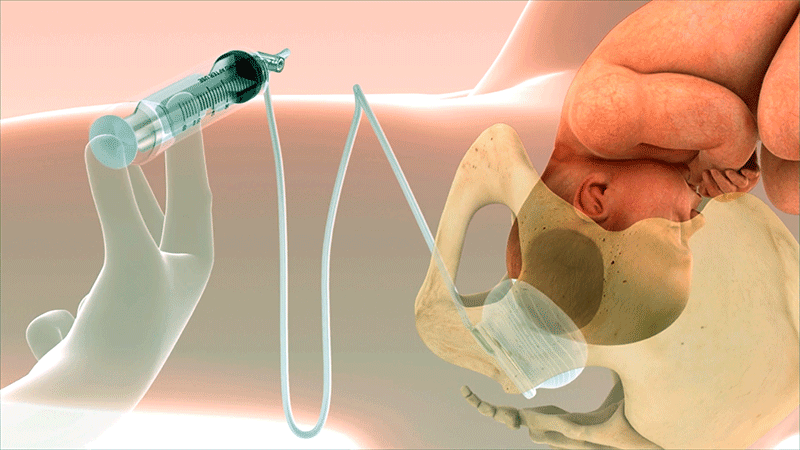
A Known Challenge in Second-Stage Cesarean Sections
Why Was Fetal Pillow Created?
Fetal Pillow was invented by Dr. Rajiv Varma to address a challenging scenario in obstetrics: cesarean delivery during the second stage of labor. Fetal Pillow provides controlled elevation of the fetal head.1
High risk, second stage cesarean section often results in increased maternal and neonatal morbidity and mortality including:
• Uterine incision extensions2,3
• Increased maternal blood loss3
• Prolonged operative time2
• Risk of fetal injury2,3
Clinically validated and FDA‑cleared as a reliable solution for challenging deliveries5
In addition to Fetal Pillow’s intended use of elevating the fetal head to help facilitate delivery, clinical benefits have been shown, including:
Reducing Maternal Complications:
- Data from a small Randomized Controlled Trial showed that Fetal Pillow™ may lower uterine extensions in a full dilation cesarean section6*
A retrospective study of 160 patients demonstrated:
- Fetal Pillow may reduce mean operative blood loss in a full dilation cesarean section7†
- Use of Fetal Pillow during cesarean delivery at full dilation may be associated with a reduction in hospital stay7‡
Reducing Fetal Complications:
- Use of Fetal Pillow in a small Randomized Controlled Trial has been shown to reduce time to delivery after hysterotomy by 23 seconds6§
- In the same retrospective study, the use of Fetal Pillow resulted in a higher mean cord arterial pH of 7.24 vs. 7.19 with hand-push method7Ω

There are more than 40 additional peer-reviewed publications evaluating the safety and efficacy of Fetal Pillow, and many of these studies have demonstrated statistically significant benefits when compared to standard techniques
Connect with our Medical Affairs team for an in-depth clinical discussion by contacting your Sales Representative
Resources for Clinicians
Hands-On Training
- In-servicing with our L&D 3-in-1 simulation trainer with your sales representative
Clinical Evidence Review
- Contact your sales representative to connect with our Medical Affairs team for an in-depth clinical discussion
Clinical Evidence Review
- Engage with experienced colleagues through Professional Education peer-to-peer programs to share best practices and real-world insights
- Contact your sales representative to connect you with CooperSurgical Professional Education
Product codes
| Product | Order Code |
|---|---|
| Fetal Pillow (6/Box) | FP-010 |

Contact your local representative for more information
IMPORTANT SAFETY INFORMATION
Fetal Pillow is indicated to elevate the fetal head and facilitate delivery of the fetus in women ≥37 weeks gestation undergoing cesarean section at full dilation (with or without an instrumental delivery). The Fetal Pillow is contraindicated in the presence of an active genital infection. The safety and effectiveness of Fetal Pillow has not been established in the following: previous cesarean section, <37 weeks gestation, non-vertex presentation, intrauterine fetal demise, pregnancy induced hypertension, intrauterine growth restriction, diabetes, major congenital abnormalities, chorioamnionitis, and multiple gestations. For detailed benefit and risk information, please consult Instructions for Use.
Footnotes
*Inflated (n = 6), Not-Inflated (n=13), numerically significant (P = 0.05).
† Fetal Pillow (n= 91), Hand-push (n= 69); mean 273 ± 145 mL vs. 403±199 mL respectively (P=0.026).
‡ Fetal Pillow (n= 91), Hand-push (n= 69); mean 77.9 ± 19.6 hours vs. 97.8 ± 27.6 hours respectively (P = 0.002).
§ Inflated (n=30), Not-Inflated (n=30); median 31 (24-37) vs. 54 (41-72) seconds respectively (P<0.01).
Ω Fetal Pillow (n= 91), Hand-push (n= 69); mean 77.9 ± 19.6 hours vs. 97.8 ± 27.6 hours respectively (P = 0.002).
References
1. Fetal Pillow – Instructions for Use
2. Van der Krogt L, Suff N, Story L, Shennana A. Management of impacted fetal head at caesarean section − Current practice and future development. Eur J of Obstet and Gyn and Repr Bio. (2025) 307:170-174.
3. Wyn Jones N, Mitchell EJ, Wakefield N, et al. Impacted fetal head during second stage Caesarean birth: A prospective observational study. Eur J Obstet Gynecol Reprod Biol. 2022;272:77-81
4. SOS website/YouTube https://youtu.be/TzfevNN4cy8?si=p1KIaWrKeii_h1_J
5. Fetal Pillow Approval Letter- August 29, 2025
6. Lassey S.C., MD, Little S.E. , et al. Cephalic Elevation Device for Second-Stage Cesarean Delivery. Obstetrics and Gynecology. 2020;00(00): 1-6
7. Safa H., Beckmann M. Comparison of maternal and neonatal outcomes from full-dilatation Cesarean deliveries using the Fetal Pillow or hand-push method.Int J Gynecol Obstet (2016), http://dx.doi.org/10.1016/j.ijgo.2016.06.013