Continued FDA Clearance
for Fetal Pillow®

We’re pleased to confirm that Fetal Pillow® cephalic elevation maintains, without interruption, an FDA 510(k) clearance. An updated review and clearance was issued in August of 2025.
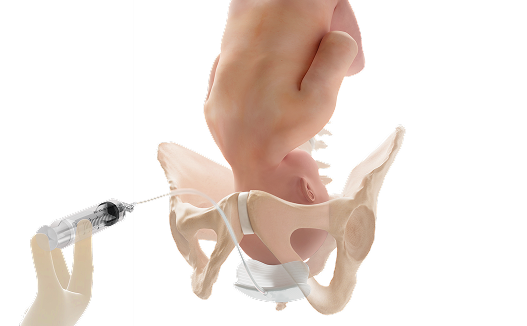
You can act with confidence knowing you have a reliable solution for challenging deliveries
Fetal Pillow performs its intended use, which is to elevate the fetal head and facilitate delivery of the fetus in women requiring cesarean section at full dilation or after a failed instrumental vaginal delivery at ≥37 weeks gestation.1
Only FDA-cleared device designed to elevate the fetal head during second-stage cesarean.
Clinical data that reinforces trust with Fetal Pillow
There are more than 40 additional peer-reviewed publications evaluating the safety and efficacy of Fetal Pillow, and many of these studies have demonstrated statistically significant benefits when compared to standard techniques. You can empower your labor and delivery teams with a tool that prioritizes patient safety and streamlines workflows.
Connect with our Medical Affairs team for in-depth clinical discussion by contacting your Sales Representative.
Impact Driven:
- As of 2025, more than 2,500 birthing hospitals globally use the device, including leading systems across the United States.
- Over 220,000 units have been purchased worldwide to streamline workflows, reduce procedure variability, and when used as directed, Fetal Pillow has a strong safety record across thousands of cases globally.
- The device has demonstrated ease of integration into existing labor and delivery workflows.
CooperSurgical is committed to advancing women’s health by providing solutions that are developed with clinical realities and patient safety in mind. Fetal Pillow was designed to integrate into established clinical workflows as an adjunctive device, supporting clinicians as they navigate challenging scenarios. In doing so, Fetal Pillow reflects CooperSurgical’s broader mission to responsibly support clinicians and patients throughout various stages of care.
Resources for Clinicians
Hands-On Training
In-servicing with our L&D 3-in-1 simulation trainer is available through your sales representative.
Clinical Evidence Review
Connect with our Medical Affairs team for in-depth clinical discussion by contacting your Sales Representative.
Peer-to-Peer Learning
- Engage with experienced colleagues through our Professional Education peer-to-peer programs, which provide opportunities to share best practices and real-world insights.
- Contact your sales representative to access CooperSurgical Professional Education resources.
- Watch our Fetal Pillow webinars.
Contact your local representative for more information
IMPORTANT SAFETY INFORMATION
Fetal Pillow is indicated to elevate the fetal head and facilitate delivery of the fetus in women ≥37 weeks gestation undergoing cesarean section at full dilation (with or without an instrumental delivery). The Fetal Pillow is contraindicated in the presence of an active genital infection. The safety and effectiveness of Fetal Pillow as not been established in the following: previous cesarean section, <37 weeks gestation, non-vertex presentation, Intrauterine Fetal Demise, Pregnancy induced hypertension, Intrauterine Growth Restriction, diabetes, major congenital abnormalities, chorioamnionitis, and multiple gestations. For detailed benefit and risk information, please consult Instructions for Use.
References

